
A fitting Drug Metabolism and Pharmacokinetic (DMPK) profile remains a challenge to reduce risks and improve efficiency in pharmaceutical R&D, leading to major drug failure cases. Compounds with optimal DMPK results seem to be efficacious and safe when choosing the same for drug development.
For the last 32 years, pharmaceutical industries have witnessed the vital role of DMPK and the science behind it, supporting and facilitating drug discovery and development process. Its predominant contribution is identifying the best drug candidates in Lead Identification and Optimization phases to enter Development phase. DMPK studies in preclinical phase can be broadly classified into in vitro DMPK and in vivo DMPK studies.
Nowadays, Electronic Laboratory Notebooks (popularly known as an ELNs) take huge credits for supporting DMPK studies. No wonder that this software system, which has been specially fabricated for scientific studies, has played a tremendous role in accelerating the preclinical studies by capturing, calculating, organizing, interacting, storing, searching and reporting scientific data across the scientific spectrum. Being a cloud-based software, it helps scientists to go paper-free.
In in vitro DMPK study there are a few sets of highly qualified assays which ensure the safety and efficacies of the drugs tested. These assays are configured as workflows in ELNs from High-throughput Screening to Lead Optimization phases. ELN assists the in vitro DMPK profile by providing a broad platform for the interaction and integrations of number of databases, instruments and other scientific setups which facilitate the process.
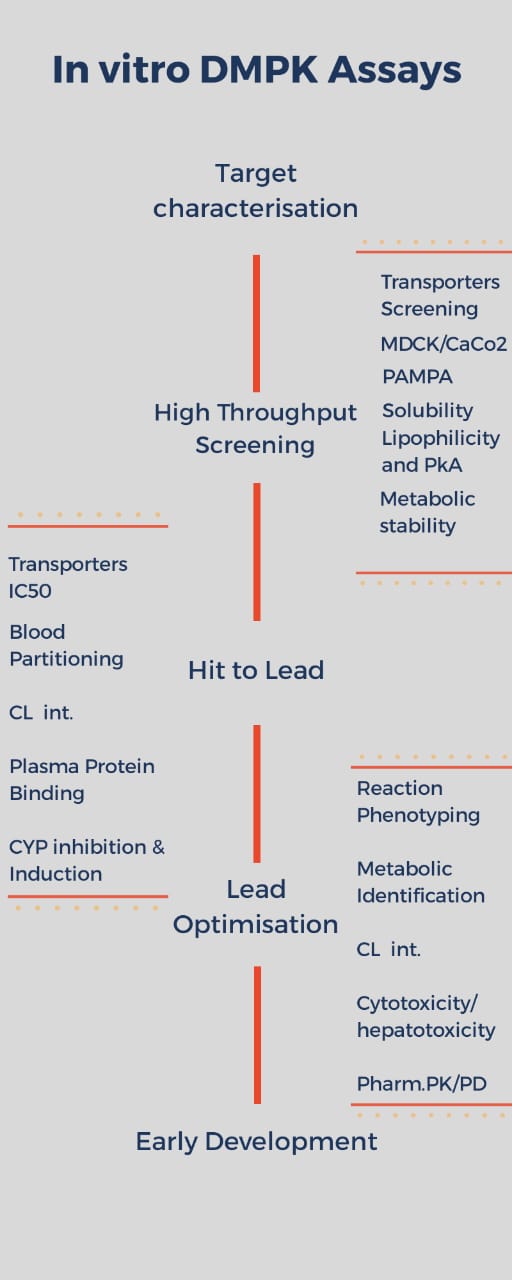
- Physicochemical screening Assays
- Purpose of the Assay
- Instruments
- Role of ELN
- Drug-transporter interaction Assays
- Purpose of the Assay
- Instruments
- Role of ELN
- Transporter screening Assays
- Purpose of the Assay
- Instruments
- Role of ELN
- Metabolic Stability Assays
- Purpose of the Assay
- Instruments
- Role of ELN
- Metabolite identification Assays
- Purpose of the Assay
- Instruments
- Role of ELN
- In-vitro CYP450 inhibition and induction screens Assays
- Purpose of the Assay
- Instruments
- Role of ELN
- Plasma Protein Binding (PPB) and Blood Partitioning Assays
- Purpose of the Assay
- Instruments
- Role of ELN
- The hepatotoxicity and cytotoxicity Assays
- Purpose of the Assay
- Instruments
- Role of ELN
WANT TO KNOW MORE?
If you would like to find out more about how Zifo can help with ELN, LIMS, Data Management, Invitro and Invivo sciences, please email us at info@zifornd.com


